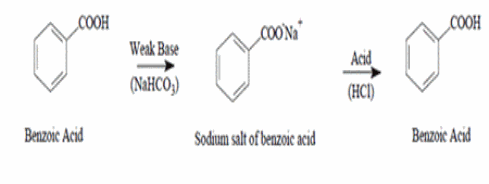
 It has a pKa of 6.3 in water which causes aqueous solutions to be mildly alkaline: HCO3- + H2O → H2CO3 + OH- The pH slightly above 7.0
It has a pKa of 6.3 in water which causes aqueous solutions to be mildly alkaline: HCO3- + H2O → H2CO3 + OH- The pH slightly above 7.0
 A carbonate ion has plenty of lone pairs.
A carbonate ion has plenty of lone pairs.
 Related Questions.
Related Questions.
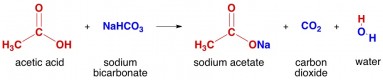
It is a base. Sodium Carbonate is a sodium salt of carbonic acid...would that make it a acid? Please explain. I have googled a few sites and they say sodium carbonate is a strong base. sodium carbonate is a basic salt. Sodium carbonate is popularly known as ash or washing soda. Well they are both salts as they are formed due to the reaction of an acid and a base.. Wiki User August 19, 2010 3:52PM. A great way to identify bases is by looking for lone pairs of electrons. Sodium bicarbonate reacts with acetic acid (found in vinegar) and presents a simple and showy demonstration of a chemical reaction. Sodium bicarbonate can indeed act as an acid. Is sodium carbonate an acid or base? When you add a hydrochloric acid (HCl) solution to a solution of sodium carbonate (Na 2 CO 3), the hydrogen ion in HCl switches places with one of the sodium ions in Na 2 CO 3 to produce sodium hydrogencarbonate, also known as sodium bicarbonate (baking soda), and sodium … It is basic in nature. On the other hand, sodium bicarbonate is represented by the formula NaHCO3 and known in layman's term as baking soda. The blood offers to the urine the identical amount of acid that it receives from the tissues and liver so it may retain its iso-form. As a strong base, sodium hydroxide neutralizes gastric acid thereby acting as an antacid. because you can prepare sodium carbonate by the reaction of sodium hydroxide and carbonic acid you will get sodium carbonate and water as this is a neutralisation reaction.sodium carbonate is basic in nature as naoh is a strong base. at school, we did a titration experiment where we titrated sodium carbonate with hydrochloric acid. But when dissolved in water sodium carbonate give a strong alkaline solution where as sodium bicarbonate is amphoteric. Sodium carbonate is a stronger base compound and is diprotic. Sodium hydrogen carbonate is a Salt of sodium ion and bicarbonate ion. Electrolysis of Aqueous Sodium Bicarbonate Using Copper To Produce Copper Carbonate. Sodium bicarbonate or sodium hydrogen carbonate is the chemical compound with the formula NaHCO3. Depending on the amount of sodium carbonate in the solution, the pH can range from around 11.3 to … Vinegar is acetic acid and is an acid. Sodium Carbonate is a base. If you react an equal number of moles of hydrochloric acid and sodium carbonate, the carbonate … Sodium carbonate is an organic sodium salt and a carbonate salt. The pH goes down in the second step as the solutes are combined to make the almost-but-not-quite strong base sodium carbonate. It has the chemical formula Na2CO3. Asked in Acids and Bases Is sodium carbonate an acid base or salt? Sodium bicarbonate or sodium hydrogen carbonate is the chemical compound with the formula NaHCO3. Is Sodium Carbonate (Na2CO3) a weak or strong base? Sodium Carbonate is the disodium salt of carbonic acid with alkalinizing property.
 It has a pKa of 6.3 in water which causes aqueous solutions to be mildly alkaline: HCO3- + H2O → H2CO3 + OH- The pH slightly above 7.0
It has a pKa of 6.3 in water which causes aqueous solutions to be mildly alkaline: HCO3- + H2O → H2CO3 + OH- The pH slightly above 7.0 A carbonate ion has plenty of lone pairs.
A carbonate ion has plenty of lone pairs. Related Questions.
Related Questions.